Cardiac surgery has evolved dramatically over the past two decades, driven by a constant effort to reduce surgical trauma while improving patient recovery and outcomes. Minimally invasive and robotic techniques now allow surgeons to perform complex procedures such as mitral valve repair, coronary artery bypass, and aortic valve surgery through smaller incisions and with less disruption to surrounding tissues. These approaches are associated with reduced postoperative pain, fewer transfusions, shorter hospital stays, and faster recovery compared with conventional sternotomy. However, this progress has introduced an important clinical paradox: while surgical access is less invasive, operative complexity often results in longer cardiopulmonary bypass (CPB) and aortic cross-clamp times.
Cardiopulmonary bypass is essential in many cardiac operations because it temporarily replaces the function of the heart and lungs, maintaining circulation and oxygenation while the heart is stopped. Yet prolonged exposure to CPB is associated with significant physiological stress. Research consistently shows that longer bypass durations correlate with higher rates of postoperative complications such as acute kidney injury, neurological impairment, systemic inflammatory response, and low cardiac output syndrome. Importantly, these risks increase disproportionately with time, particularly once certain thresholds—often around 90 minutes of cross-clamp time—are exceeded.
This relationship between time and risk is particularly concerning in elderly or frail patients. Frailty represents a complex physiological state characterized by reduced metabolic reserve, impaired microcirculation, chronic inflammation, and diminished ability to tolerate surgical stress. In such patients, even relatively small increases in bypass time can significantly increase postoperative morbidity and mortality. Traditional preoperative frailty scores provide only a static snapshot of risk and do not account for the dynamic physiological responses that occur during surgery.
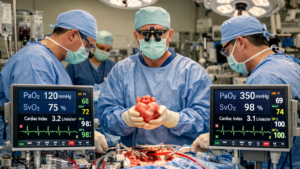
The article proposes that the solution to this paradox lies in the digital transformation of perfusion management. Historically, perfusion monitoring relied on intermittent measurements such as arterial pressure, venous oxygen saturation (SvO₂), and occasional lactate sampling. These metrics provide useful information but offer only indirect insight into tissue perfusion and metabolic stress. Because they are measured intermittently, they often reveal physiological problems only after they have already developed.
Digital perfusion systems fundamentally change this paradigm. Modern heart–lung machines can integrate high-frequency sensor data, capturing thousands of physiological data points per minute. Parameters such as oxygen delivery (DO₂), carbon dioxide production (VCO₂), blood flow, hematocrit, temperature, and pressure are continuously recorded and displayed through integrated digital dashboards. This shift from intermittent monitoring to continuous data streams allows clinicians to identify emerging perfusion problems earlier and intervene before irreversible organ injury occurs.
Beyond monitoring, digital systems enable predictive analytics using machine learning algorithms. By analyzing complex relationships between perfusion variables and patient physiology, these algorithms can estimate the probability of complications such as acute kidney injury in real time. Rather than simply reacting to abnormal values, perfusionists and surgeons can receive alerts indicating that a patient’s physiological trajectory is trending toward risk. For example, sustained periods of low indexed oxygen delivery (DO₂i) during CPB have been shown to correlate strongly with postoperative kidney injury. Predictive models can detect these harmful trends early and recommend adjustments such as increasing pump flow or perfusion pressure.
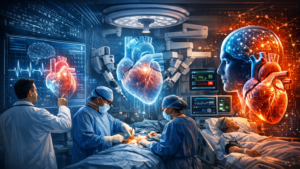
One of the most promising innovations discussed in the article is the concept of the “digital twin.” A digital twin is a computational model representing an individual patient’s cardiovascular and metabolic system. By integrating continuous intraoperative data, the digital twin can simulate how the patient’s physiology will respond to different perfusion strategies. This allows clinicians to test potential interventions virtually before applying them to the patient, supporting personalized and anticipatory decision-making during surgery.
The digital evolution of perfusion management can be understood as a progression through three stages. The first stage is continuous monitoring, where raw physiological signals are captured at high frequency. The second stage is prediction, where machine learning models transform these signals into dynamic risk estimates. The third stage is decision support, where predictive analytics provide automated alerts or adaptive control recommendations for pump flow, temperature management, and gas exchange.
However, the success of these technologies depends on reliable and standardized data infrastructure. Historically, perfusion data have been fragmented across multiple devices and proprietary systems, limiting interoperability and preventing large-scale analysis. Establishing standardized datasets and integrating perfusion consoles with electronic health records and Internet-of-Things sensors will be essential for enabling predictive algorithms to learn from diverse patient populations.
The authors also emphasize that digital transformation must be accompanied by ethical oversight and responsible implementation. Artificial intelligence systems used in healthcare must remain transparent, explainable, and subject to clinical supervision. Data governance and cybersecurity protections are also critical to safeguard patient privacy.
Despite these challenges, the integration of digital health technologies into cardiac surgery represents a major opportunity to redefine how clinicians think about cardiopulmonary bypass. Traditionally, bypass duration has been treated as an unavoidable procedural metric. In the digital era, it may instead become a dynamic physiological variable that can be optimized in real time.
Ultimately, the article argues that precision perfusion—guided by continuous monitoring, predictive analytics, and digital twin modeling—could help reconcile the benefits of minimally invasive cardiac surgery with the need to maintain physiological safety. By transforming CPB management from reactive to predictive, digital technologies may reduce time-dependent complications and extend the duration of safe bypass, improving outcomes for patients undergoing modern cardiac surgery.