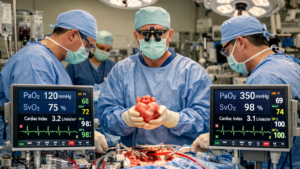
The publication titled Cardiopulmonary Bypass as Safe and Effective Support for Concomitant High-Risk Percutaneous Coronary Intervention and Transcatheter Aortic Valve Replacement addresses a critical and increasingly encountered clinical scenario in interventional cardiology: patients with severe aortic stenosis and complex multivessel coronary artery disease who require both revascularization and valve intervention. As the population ages and transcatheter therapies expand, clinicians are frequently faced with individuals who are too high risk for conventional surgical aortic valve replacement combined with coronary artery bypass grafting, yet remain hemodynamically fragile during percutaneous approaches. This study explores the role of cardiopulmonary bypass (CPB) as a mechanical circulatory support strategy to enable safe and effective completion of combined high-risk PCI and TAVR procedures.
Patients presenting with severe aortic stenosis often have coexisting obstructive coronary artery disease. In many cases, complete revascularization prior to or during TAVR is desirable to optimize myocardial perfusion and reduce peri-procedural ischemic complications. However, high-risk PCI—particularly in the setting of left main disease, reduced left ventricular ejection fraction, complex bifurcation lesions, or chronic total occlusions—can lead to hemodynamic instability. When combined with the rapid pacing and transient hypotension required during TAVR valve deployment, the cumulative physiologic stress may exceed the patient’s compensatory reserve. The authors propose that planned use of CPB provides full circulatory support, ensuring end-organ perfusion and myocardial stability throughout these prolonged and complex procedures.
The study describes a cohort of patients undergoing concomitant high-risk PCI and TAVR with CPB support. These individuals were characterized by advanced age, multiple comorbidities, significant coronary disease burden, and severe symptomatic aortic stenosis. Many were deemed poor surgical candidates due to elevated operative risk scores and frailty. The decision to employ CPB was made pre-procedurally by a multidisciplinary heart team, emphasizing the importance of collaborative structural heart programs in managing such complex cases.
Cardiopulmonary bypass was initiated to provide systemic perfusion and ventricular unloading during coronary intervention and valve deployment. This strategy allowed operators to perform complete and meticulous PCI without the constraint of impending hemodynamic collapse. Complex lesions were treated with greater procedural confidence, and the risk of ischemia during balloon inflation or stent placement was mitigated by maintained circulatory flow. Following revascularization, TAVR was performed under continued hemodynamic support, reducing the risk associated with rapid ventricular pacing and valve positioning.
The reported outcomes demonstrate that CPB can be implemented safely in this hybrid interventional setting. Procedural success rates were high, with successful completion of both PCI and TAVR in the majority of cases. Hemodynamic parameters remained stable during intervention, and conversion to emergent surgical intervention was not required. Complication rates, including bleeding, vascular injury, stroke, or acute kidney injury, were within acceptable ranges given the high-risk nature of the population. Importantly, early survival outcomes were favorable, suggesting that CPB did not introduce prohibitive additional risk.
One of the key advantages highlighted by the study is the ability to achieve more complete revascularization. In hemodynamically unsupported high-risk PCI, operators may limit intervention due to hypotension or instability, potentially resulting in incomplete treatment of significant lesions. With CPB support, the interventional team could pursue a more comprehensive strategy, which may translate into improved long-term myocardial function and symptom relief. Additionally, ventricular unloading during CPB may reduce myocardial oxygen demand and limit ischemic injury during prolonged procedures.
The authors also discuss alternative mechanical circulatory support devices, such as intra-aortic balloon pump (IABP), Impella, or extracorporeal membrane oxygenation (ECMO). While these devices provide varying degrees of hemodynamic support, CPB offers full cardiopulmonary support with robust flow rates and oxygenation capacity. In the context of simultaneous complex coronary and valvular intervention, this level of support may offer unique advantages. However, CPB is more invasive and requires surgical cannulation expertise, underscoring the need for institutional experience and a coordinated heart team.
Despite its promising findings, the study has limitations. The sample size is relatively small, and the design is observational rather than randomized. There is no direct comparison group of patients undergoing combined PCI and TAVR without CPB support, limiting definitive conclusions about superiority. Selection bias is also possible, as patients were carefully chosen for this strategy based on clinical judgment. Nonetheless, the results contribute meaningful real-world data to a growing body of literature supporting mechanical circulatory support in high-risk structural and coronary interventions.
In conclusion, this study provides evidence that cardiopulmonary bypass can serve as a safe and effective hemodynamic support strategy during concomitant high-risk PCI and TAVR in carefully selected patients. By stabilizing circulation, enabling complete revascularization, and mitigating procedural risk, CPB expands the therapeutic options for individuals who might otherwise face limited treatment pathways. As transcatheter therapies continue to evolve, integration of advanced mechanical support techniques within multidisciplinary structural heart programs will likely play an increasingly important role in managing the most complex cardiovascular patients.