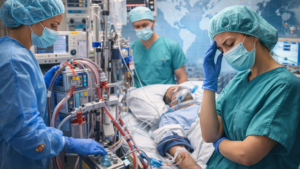
The Invisible Face of ECMO: Anxiety Levels and Challenges Among Intensive Care Nurses
This cross-sectional study examined 137 ICU nurses in Turkey who cared for patients receiving ECMO, a complex life-support therapy. Nurses reported mild-to-moderate anxiety, with higher stress linked to bleeding risk, infection concerns, workload, complications, and team-related organizational problems. More ECMO-specific experience was associated with lower anxiety, highlighting the value of structured training, standardized protocols, and experienced staffing.